 
Moreover, one can measure their speed exactly along with their locations. 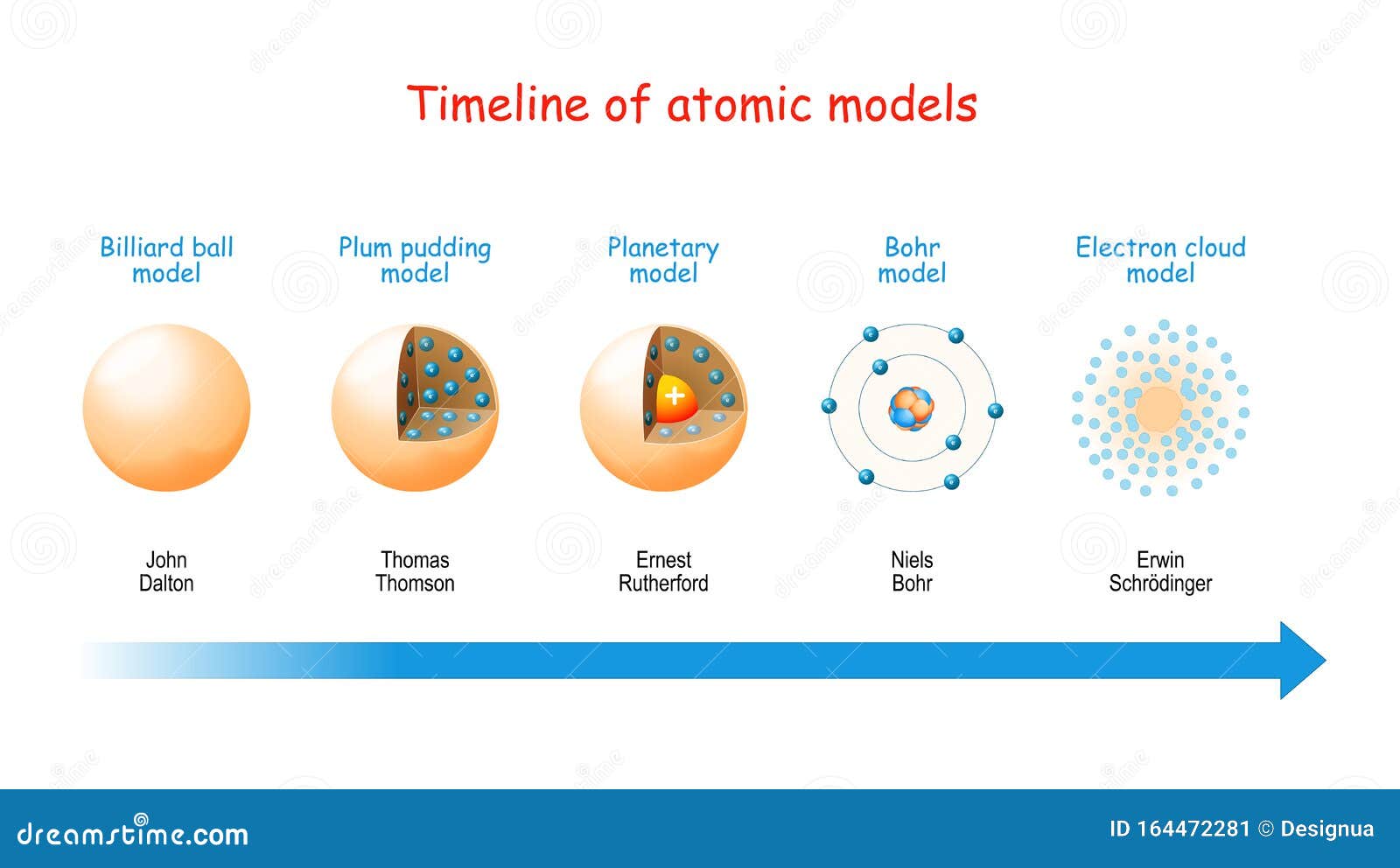
On the other hand, modern quantum theory specifies them as statistical “clouds”. Earlier theories used to treat electrons, and other tiny particles as fixed solid “lumps,”. 6. Einstein, Heisenberg and Quantum MechanicsĪs far as the earlier theories are concerned, the atom consists of a central and heavy nucleus centered by a number of electrons. At the time of returning to their original orbit, they leave this energy as electromagnetic radiation. When these atoms grasp the energy and move into a higher orbit, this theory refers to them as “excited” electrons. When the electrons are in orbit, they possess “constant energy.” In 1913, Danish physicist Niels Bohr proposed a planetary model, which states that electrons revolve about the nucleus just as the planets orbit the sun. He further hypothesized that the number of protons and electrons are equals in an atom. In the past, he discovered the part of activity such as the movement of protons and electrons within the central part of the atom. In 1911, Ernest Rutherford (British physicist) proposed a nuclear model on atoms. Today we call the positive charged particles protons and the negative one’s electrons. He does so after discovering electrons in 1897.Īlso, his model suggested that atoms consist of a big positively-charged sphere studded with negatively charged electrons (he called them “corpuscles”) like fruit in a plum pudding.įurthermore, he put forward that the charge of the positive sphere’s charge is equal to the negative charges of the electrons. Thomson proposes the “plum pudding” theory of the divisible atom. He also proposed that while all atoms of one element are identical, they are totally different from those that make up other elements. He postulated that matter is made of atoms, which are small indivisible particles. Dalton’s Atomic TheoryĮnglish chemist John Dalton subsequently made on the Greek notion of atoms in 1808. Moreover, they also made some intangible qualities such as taste and color. In addition, they tell that these were solid particles without internal structure, and came in a variety of shapes and sizes. 
“All the matter is made up of tiny units called atoms” this was first proposed by Leucippus and Democritus, in the fifth century B.C., that all matter is made of tiny units called atoms. The total mass of all products of a chemical reaction is equal to the total mass of all reactants of that reaction.List of Atomic Theories 1. Ancient Greek Beliefs Finally, we all assume that we have demonstrated the Law of Conservation of Mass. 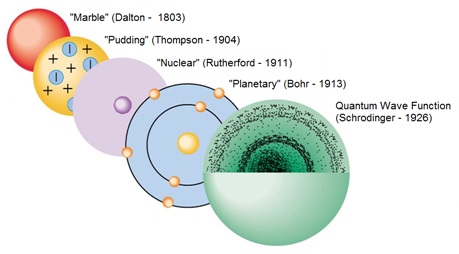
The elements are not transmutable: one element cannot be converted into another. For example, metallic iron and gaseous oxygen are both elements and cannot be reduced into simpler substances, but iron rust, or ferrous oxide, is a compound which can be reduced to elemental iron and oxygen. All other pure substances, which we call compounds, are made up from these elements and can be decomposed into these elements. We will assume that we have identified all of these elements, and that there are a very small number of them. We will begin by assuming that all materials are made from elements, materials which cannot be decomposed into simpler substances. There are over 18 million known substances in our world. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
